
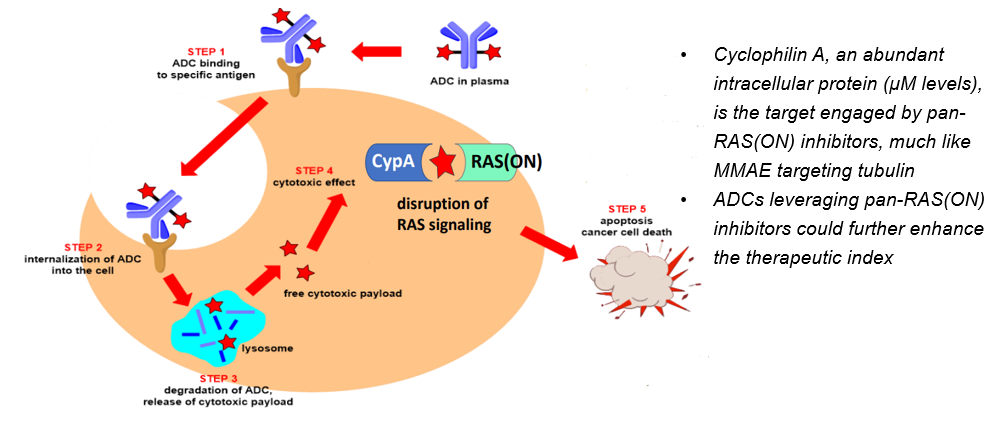
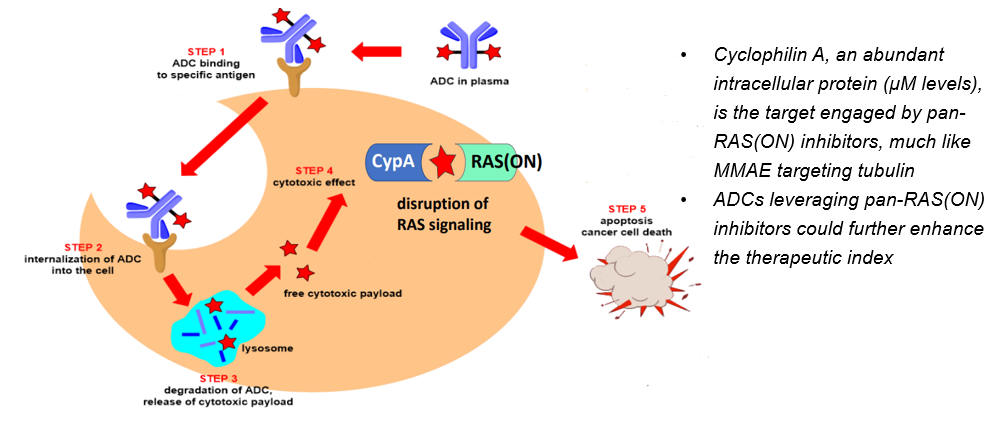
Hypothesis: Targeted delivery of a pan-RAS(ON) inhibitor in the form of ADC could widen its therapeutic index and enable rational combinations
• Addressing the limitations of small molecule pan-RAS(ON) inhibitors
– Tolerability issues: While shown to be safe, RMC-6236 has tolerability issues at the RP2D (300 mg QD for PDAC & 200 mg QD for NSCLC). Escalation beyond the MTD to further improve efficacy is not tolerable
– Combination challenges: Combination therapy with an EGFR monoclonal antibody in colorectal cancer (CRC) may be limited by overlapping dermatologic toxicity. In addition, combining with standard-of-care (SOC) chemotherapy may present tolerability challenges
• Adlai Nortye is positioned to develop first-in-class RASi ADC with transformational potential
– The picomolar to sub-nanomolar potency of our internal library of pan-RAS(ON) inhibitors, together with the high intracellular abundance of cyclophilin A, supports their potential as ideal ADC payloads for both RAS-mutant and selected RAS wild-type tumors
– Strong IP position for Linker-Payload
– Existing expertise accumulated from novel ADC payload programs, as well as significant translational/clinical experience in ADC development
AN4035 is the 1st drug candidate to demonstrate Proof-of-Concept of the RASiCATM platform by showing compelling preclinical efficacy and tolerability in CEACAM5-expressing RAS mutant tumors as monotherapy or in combination. IND submission is anticipated in mid-2026.